Publications
Find coverage of the latest original articles on Lupus, focusing on those with data on therapeutic interventions and those that have clinical impact.
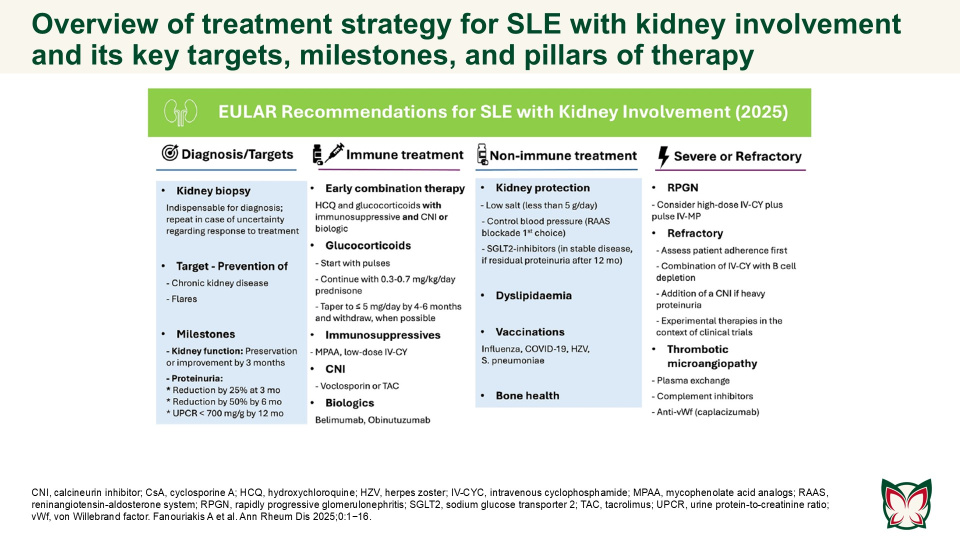
EULAR recommendations for the management of systemic lupus erythematosus with kidney involvement: 2025 update
Ann Rheum Dis 2025;0:1−16 Doi: 10.1016/j.ard.2025.09.007
The updated EULAR recommendations provide evidence- and expert-based consensus on the management of SLE with kidney involvement, adjusted for severity, and taking into consideration long-term efficacy, safety, cost, and local availability of drugs. Fanouriakis A et al. updated the 2019 EULAR/ ERA-EDTA recommendations for the management of SLE with kidney involvement, taking into consideration emerging evidence and recent developments in the field.
Keywords:
Safety and Efficacy of Long-term Voclosporin Treatment for Lupus Nephritis in the Phase 3 AURORA 2 Clinical Trial
Arthritis Rheumatol. 2023;76(1):59–67 DOI: 10.1002/art.42657
AURORA 2 demonstrated the safety and tolerability of continued administration of voclosporin over 3 years of treatment in patients with LN.
Keywords:
Update on the Efficacy and Safety Profile of Voclosporin: An Integrated Analysis of Clinical Trials in Lupus Nephritis
Arthritis Care Res (Hoboken). 2023 Jul;75(7):1399–1408 DOI: 10.1002/acr.25007
Pooled analysis of data from the AURA-LV phase 2 and AURORA 1 phase 3 trials of voclosporin in patients with active LN demonstrated that significantly more patients achieved a complete renal response at 1 year in the voclosporin than the control group (p<0.0001), with no observation of new safety signals.
Safety and Efficacy of Belimumab in Patients with Lupus Nephritis
Clin J Am Soc Nephrol. 2022;17:1620–1630 doi: 10.2215/CJN.02520322
This 28-week, open-label extension of BLISS-LN found no new safety signals and maintained efficacy with belimumab, in patients with lupus nephritis.
Keywords:
A secondary analysis of the Belimumab International Study in Lupus Nephritis trial examined effects of belimumab on kidney outcomes and preservation of kidney function in patients with lupus nephritis
Kidney Int. 2022;101(2):403-413 doi: 10.1016/j.kint.2021.08.027
Post-hoc analysis data suggests that the addition of belimumab to standard therapy may be effective in preserving long-term kidney function in patients with lupus nephritis (LN).