Publications
Find coverage of the latest original articles on Lupus, focusing on those with data on therapeutic interventions and those that have clinical impact.
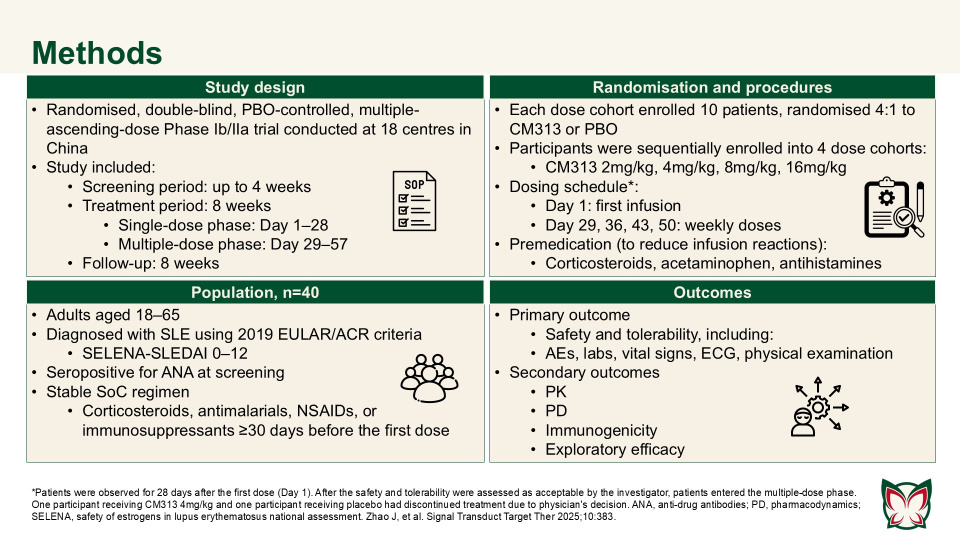
Anti-CD38 monoclonal antibody CM313 for systemic lupus erythematosus: A randomized, double-blind, placebo-controlled Phase Ib/IIa trial
Signal Transduct Target Ther 2025;10:383 Doi: 10.1038/s41392-025-02487-2
Zhao et al. showed that CM313 was well tolerated in adult patients with SLE at doses of 2–16mg/kg and showed encouraging pharmacodynamic effects and preliminary efficacy at doses of 8 and 16 mg/kg QW. CM313 also produced dose-dependent and clinically meaningful improvements in key serological biomarkers of SLE.
Efficacy and safety of allogeneic CD19 CAR NK-cell therapy in systemic lupus erythematosus: A case series in China
Lancet 2025 Doi:10.1016/S0140-6736(25)01671-X Epub ahead of print
Gao et al. report that allogeneic CAR NK-cell therapy is a potent option for treatment of autoimmune diseases and may address limitations of current autologous CAR T-cell therapy, including manufacturing scale and time, access, safety, and cost. Authors evaluated the safety, tolerability, and efficacy of allogeneic CD19 CAR NK-cell therapy in patients with relapsed or refractory SLE.
Keywords:
Unsupervised machine learning identifies distinct systemic lupus erythematosus patient endotypes with differential response to belimumab
Rheumatol (Oxford), 2025 DOI: 10.1093/rheumatology/keaf215. Epub ahead of print
Depascale et al. used unsupervised machine learning to identify three SLE endotypes based on B cell immunophenotyping and serological profiles. Belimumab was most effective in patients with transitional and naïve B cell enrichment (Cluster 2), where it significantly increased the likelihood of achieving sustained LLDAS and DORIS remission.
Keywords:
Opportunities and limitations of B bell depletion approaches in SLE
Nature Review Rheumatol, 2025;21:111–126 DOI: 10.1038/s41584-024-01210-9
Stockfelt et al. reviewed the long-term efficacy and challenges of B cell depletion strategies in SLE. Rituximab, a CD20-targeting monoclonal antibody, has demonstrated efficacy in a subset of patients but remains limited by immunogenicity, residual B cells, and B-cell activating factor (BAFF)-mediated relapse. Newer strategies incorporating CAR T cells, bispecific T cell engagers, and combination therapies aim to enhance B cell depletion and optimise outcomes.
Keywords:
Efficacy and safety of obinutuzumab in active lupus nephritis
NEJM, 2025. Epub ahead of print. DOI: 10.1056/NEJMoa2410965
Furie et al. demonstrated that obinutuzumab plus standard therapy significantly improved complete renal response at Wk76 compared with placebo. No unexpected safety signals were identified, though infections and COVID-19-related events were more frequent in the obinutuzumab group.
Clinical and biomarker responses to BI 655064, an antagonistic anti-CD40 antibody, in patients with active lupus nephritis: a randomized, double-blind, placebo-controlled, phase II trial
Arthritis Rheumatol. 2023;75(11):1983–93 doi: 10.1002/art.42557.
This phase II study of BI 655064 in patients with active LN did not meet the primary endpoint of CRR at Week 52, however, post-hoc analyses suggest a potential benefit of BI 655064 180 mg in patients with active LN.
Machine Learning Identifies Clusters of Longitudinal Autoantibody Profiles Predictive of Systemic Lupus Erythematosus Disease Outcomes
Ann Rheum Dis 2023;82(7):927–36 doi 10.1136/ard-2022-223808
Choi, et al. used machine clustering techniques to divide SLE patients into four distinct clusters. This could potentially be used to predict future clinical outcomes, and as benchmarks to study other SLE-related outcomes.
Keywords:
Efficacy and Safety of the Bruton's Tyrosine Kinase Inhibitor Evobrutinib in Systemic Lupus Erythematosus: Results of a Phase II, Randomized, Double-Blind, Placebo-Controlled Dose-Ranging Trial
ACR Open Rheumatol. 2023;5(1):38–48 doi: 10.1002/acr2.11511
Results of a phase II trial of evobrutinib suggest that Bruton's Tyrosine Kinase (BTK) inhibition is not an efficacious therapeutic intervention over standard of care therapy for patients with SLE.
Keywords:
Trial of Anti-BDCA2 Antibody Litifilimab for Systemic Lupus Erythematosus
N Engl J Med. 2022;387(10):894–904 doi: 10.1056/NEJMoa2118025
Phase 2 study, in patients with systemic lupus erythematosus, shows that litifilimab is associated with a greater reduction from baseline in the number of swollen and tender joints than placebo, over a period of 24 weeks.
Phase II randomised trial of type I interferon inhibitor anifrolumab in patients with active lupus nephritis
Ann Rheum Dis. 2022;81(4):496–506 doi: 10.1136/annrheumdis-2021-221478
Despite not meeting the primary endpoint, this Phase II trial of anifrolumab in patients with active lupus nephritis (LN) demonstrates that anifrolumab IR is associated with numerical improvements over placebo across endpoints – including complete renal response – in patients with active LN.